Particles can cause irritation to the eyes,
nose and throat. Some of the larger particles (> 10 µm
diameter) reaching the nose or throat will be filtered out by the
body's natural defense system. However, very tiny particles that
reach deep into the lung may be absorbed into the blood stream or
cause lung or other health problems. Such particles are those
less than 10 µm in diameter - hence the term PM10.
Studies in the United States have shown that death rates from
respiratory and cardiovascular disease increase with increased
concentrations of PM10, and EU monitoring shows
significant levels in many countries.
Both the size and composition of the
particles have potential health effects. In the winter smogs of
earlier this century, it was soot and SO2 that were
identified as a health hazard, responsible for bronchitis and
emphysema. These acidic particles can cause lung inflammation and
changes in blood clotting. No direct link has been proved between
particulate pollution and asthma.
 Ozone
Ozone
There are two sorts of ozone: ozone in the
stratosphere (15-50 km above the earth's surface) forms what is
known as the "ozone layer" and is essential in limiting
the amount of ultraviolet irradiation reaching the earth's
surface (see section on global- effects, p. a19). However, ozone
in the troposphere - the level that contains the air we breathe -
is a pollutant and it can damage health and vegetation.
At ground level ozone is a secondary
pollutant(2) formed by the
action of sunlight on primary pollutants; these are nitrogen
oxides from vehicle emissions and industry and volatile organic
compounds from vehicles, solvents and industry. Nitrogen oxides
react in sunlight (photochemical reaction) to form ozone, a major
constituent of photochemical smog.
High concentrations of ground level ozone
are often a particular problem in hot sunny climates such as in
southern Europe, with cities like Athens having a
particular problem. In northern Europe concentrations are usually
higher in rural areas as ozone production occurs in polluted air
as it drifts away from cities. Ozone is thus also a transboundary
pollution problem and action to deal with it needs to be agreed
nationally and internationally and implemented locally.
While ozone can damage crops and
vegetation, its activity is such that concentrations indoors are
rapidly reduced by reaction with plastics and fabrics; almost
alone amongst the major air pollutants, exposure to ozone is
nearly always an outdoor problem.
| Asthma |
| Asthma
is a disorder of the airways in which inflammation
obstructs airflow, causing breathlessness and wheezing.
It is becoming increasingly common throughout the
developed world in both urban and rural areas. It is an
inflammatory disease which causes the bronchi to
overreact to a stimulus. To counteract the irritant, the
airways become inflamed and swollen and produce excess
mucus causing narrowing of the airways in vulnerable
individuals. In the UK asthma is thought to affect
between 4 - 6% of children and 4% of adults, when in
Finland, over 2.5% of the population suffers from asthma.
A common feature through all Western countries is a
strong increase in the number of asthma cases. See also
WHO pamphlet on Asthma. |
The biological response to ozone is
dependent upon the concentration to which an individual is
exposed and the duration of exposure; the dose received is also
dependent upon the volume of air inhaled per minute as nearly all
the ozone inhaled is absorbed. Thus people taking strenuous
physical exercise, e.g. jogging, inhale more ozone and are likely
to show a proportionally greater response to its effects. The
biological response to ozone is unusual though not unique. If
exposure is continued over several days, the response falls away
and the person is described as having developed tolerance to
ozone. This phenomenon has not been adequately explained and
decrease in response should not be taken as an indication that
exposure day after day does no more harm than a single day's
exposure.
The importance of duration of exposure, and
the fact that this may extend over 8 hours on a sunny day, has
led to guidelines and air quality standards for ozone usually
being defined in terms of an 8-hour average concentration. This
is commonly exceeded in Europe.
In terms of producing inflammation of the
respiratory tract, ozone is one of the most toxic of the common
air pollutants. According to WHO hourly concentrations of 200
µg/m3 can cause eye, nose and throat irritation,
chest discomfort, cough and headache; exposure for about six
hours to concentrations of 160 µg/m3 have been shown
to produce inflammation of the airways and changes in standard
indices of lung function.
Individuals show an enormous variation in
their response to ozone, with asthma sufferers not being
significantly more affected than others; there is however limited
evidence to suggest that any ill health effects may be longer
lasting in asthmatics.
Ozone has also been shown to increase the
response, or to increase the sensitivity, of some people when
exposed to the substance to which they are allergic. This has
been confirmed in several volunteer studies and in an
epidemiological study of hay fever sufferers in London. The fact
that peak levels of pollen and of ozone often occur together may
make this an important observation.
 Nitrogen Dioxide
Nitrogen Dioxide
Nitrogen dioxide (NO2) is
produced both as a primary and as a secondary pollutant by
combustion processes. In many countries some 50% of NO2
is produced by motor vehicles. Thus concentrations tend to be
higher in busy streets than in rural areas.
Concentrations of NO2 show a
distinct daily variation with peak levels typically being
recorded during the morning and evening rush hours. In cold,
still weather concentrations may increase due to trapping of
pollutants in a layer of wet air close to the ground. These
episodic increases in concentrations of NO2 do not
reflect a change in sources but a change in the pattern of
dispersion of the pollutants. The atmospheric conditions which
cause an increase in concentrations of NO2 also cause
an increase in concentrations of other pollutants, such as
particles and carbon monoxide.
Nitrogen dioxide is an irritant gas and
exposure to exceedingly high concentrations produces narrowing of
the airways in both asthmatic and non-asthmatic individuals.
Asthmatics are more sensitive to NO2 than
non-asthmatics with exposure to concentrations of about 560 µg/m3
for 30 minutes producing a small change in standard indices of
lung function; in non-asthmatics exposure to about 1800 µg/m3
would be necessary to produce a similar response. The
exposure-response relationship for NO2 is erratic.
Exposure to concentrations of 560 µg/m3 may produce a
response whilst exposure to double that may not; the response
does seem to reappear and remain as concentrations approach and
exceed 1800 µg/m3. The explanation for this is not
clear.
| Smog in London,
December 1991 |
| A
dramatic example of the effect that weather can have on
the dispersal of pollutants occurred in London in
December 1991: from the morning of 12 December until the
evening of 15 December, London was blanketed in its worst
smog since 1952. During those four days nitrogen dioxide
concentrations were well above 190 µg/m3 for
most of the time, peaking at 800 µg/m3 in
South West London on Friday 13 December. Up to 160
additional deaths are thought to have occurred during
this period. Deaths from respiratory disease (including
asthma) were 22% higher and from cardiovascular disease
14% higher than expected when compared with the same
period the previous year. |
Children living in homes using gas cookers
have been shown to have an increased risk of respiratory
infections compared to those living in homes with electric
cookers. This has been attributed to long-term exposure to raised
concentrations of NO2 although the evidence is not
conclusive. Recent studies have also shown that long-term
exposure to the pollutants generated by gas cookers is associated
with increased symptoms of asthma amongst women. This effect has
also previously been attributed to NO2, but again the
evidence is inconclusive. (See pamphlet Indoor air quality.)
Exposure to NO2 at
concentrations not very much in excess of those occasionally
experienced both indoors and out has also been shown to increase
the response of sensitive individuals to allergens. The
importance of this effect on public health is uncertain though
some, but not all studies, have suggested an increased prevalence
of asthma in those living close to busy roads. Further research
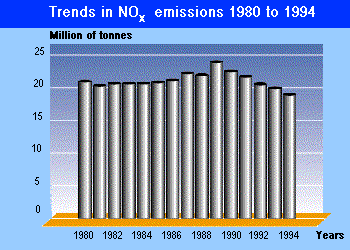
in this area is required. Emissions of total nitrogen oxides,
including NO2 are also declining slowly, but remain
above acceptable levels.
 Carbon Monoxide
Carbon Monoxide
Carbon monoxide (CO) has an affinity for
haemoglobin (Hb), the substance that carries oxygen around our
bodies in our blood, 200 times greater than that of oxygen. It
impairs the oxygen-carrying capacity of blood as
carboxyhaemoglobin (COHb) is formed rather than oxyhaemoglobin.
When too much oxygen is displaced by CO, it can progressively
lead to oxygen starvation. Thus at levels of only a few percent
COHb effects may be significant in tissues which are already
deprived of oxygen, perhaps as a result of poor blood supply. The
escalating symptoms of CO poisoning are headache and vomiting
and, in severe cases, collapse and death, although the effects of
brief exposure are reversible.
Cigarette smokers seem to adapt to high
levels of COHb: often as much as 10% of total haemoglobin is
combined with CO as COHb in the blood of heavy cigarette smokers.
Indeed, cigarette smokers are on average net contributors to
ambient CO levels.
In healthy, non-smoking, individuals
effects of exposure to CO appear at a COHb concentration of about
5%. In those with poor circulation to the heart it has been shown
that levels of COHb as low as 2.5% may produce an effect upon the
electrical activity of the heart.
Carbon monoxide is a problem both as an
indoor and as an outdoor air pollutant. In terms of accidental
deaths, indoor exposure presents the major problem. Despite the
undoubted and well understood toxic effects of CO there was until
recently little evidence to suggest that outdoor exposure posed a
risk to health. However, recent studies have shown that the
increase in emergency admissions to hospital for heart attacks,
which had been reported as being related to changes in levels of
particles, was also significantly related to changing levels of
CO. This suggests that exposure to low concentrations of CO may
be dangerous and that rather more deaths than previously thought
may occur from outdoor exposure to CO.
 Lead
Lead
Lead is a heavy metal which exists
naturally in the earth's crust. It is found throughout the
environment in water and air. Natural concentrations of lead are
low but exploitation of lead as a useful metal has increased the
level of exposure to this contaminant. In those countries which
still mainly use leaded petrol, most of the lead in the air comes
from petrol-fuelled vehicles. While the dangers of exposure to
high levels of lead are well known, there is very serious concern
that low levels of lead may affect the mental development of
children (see WHO pamphlet on Lead and
Health).
 Carcinogens as Outdoor Air Pollutants
Carcinogens as Outdoor Air Pollutants
Association and causality
Source: Handbook on air
pollution and health
|
| It is often
fairly easy to show that measure of ill-health, e.g. the
number of admissions to hospital per day, is associated
with a possible cause such as the day-to-day variation in
levels of air pollutants. To show real cause and effect,
i.e. that a causal relationship exists, a number of
guidelines or tests have been developed. These include
looking at the consistency of the results of a number of
different studies, the way in which the results of
different studies fir together (coherence), whether there
is a "dose-response relationship" such as that
as the proposed causal factor increases so does the
effect and whether the sequence of events makes sense,
i.e. the cause always precedes the effect. Proof
of causality is often impossible but by the application
of these and other criteria an expert judgment as to
whether an association is likely to be causal can often
be reached.
|
A considerable number of substances,
carcinogenic or supposedly carcinogenic, occur as outdoor and
indoor air pollutants. Of these benzene, 1,3-butadiene and the
polycyclic aromatic hydrocarbon compounds (PAHs) are perhaps the
best known. Some of them are genotoxic carcinogens and as such
any level of exposure (no matter how small) may, in theory, be
associated with an increased risk of cancer. The term
"cancer" is used here to embrace all malignant tumours
and related disorders such as leukemia. Attempts to quantify the
risk posed by exposure to ambient levels of carcinogens have been
made though the assumptions underlying these calculations have
been challenged.
For air pollutants which are considered to
be genotoxic carcinogens, WHO experts have used mathematical
models to calculate so called "unit risk". These
calculations are based on information from animal studies or from
epidemiological studies. This unit risk is generally expressed as
the additional cancer risk in a given population that inhales
this compound at a concentration of 1 µg/m3 during its entire
lifetime. For benzene, for instance, the unit risk factor is
4.10-6. This means that in a population of one million persons, 4
additional cases of cancer can be expected following a continuous
inhalation of 1 µg/m3 during its lifetime or one additional
cancer in a population of 250 000 people.
It should be realized, however, that these
theoretical risk estimates contain large safety factors, but also
a number of uncertainties.
To derive standards, countries have to
choose their acceptable level of risk for each hazard, balancing
risks and benefits, and establishing the degree of urgency of
public health problems among sub-populations inadvertently
exposed to carcinogens. In the United Kingdom, the standard for
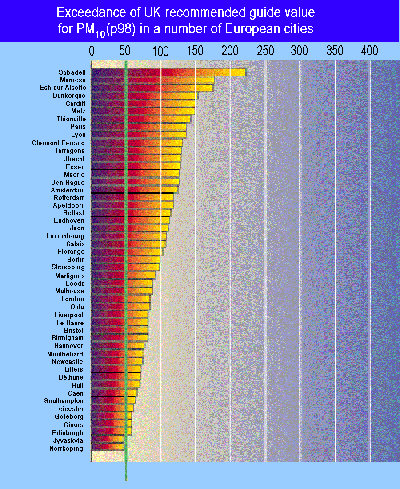
benzene is 5 µg/m3. This level is exceeded in many instances
(see table above) in many cities, but one should keep in mind
when comparing measured concentrations in the air to a guideline
value that:
- models used to derive the guideline
values are likely to be conservative; and
- exposure is rarely continuous.
Nevertheless, efforts should be made to
reduce the level of carcinogens in the air.
 Indoor Air Pollutants
Indoor Air Pollutants
As well as nitrogen dioxide, carbon
monoxide and lead there are a number of other pollutants that
affect air quality in enclosed or confined spaces. More detailed
information is available in the
pamphlet indoor air quality as well as in other pamphlets such as
Radon, Asbestos and health or
Asthma and respiratory allergies.
Clearly, indoor air pollution affects people both in their homes
and at the workplace. In the latter case, workers cannot choose
the air they breathe and there is a need for legislative or
regulatory action. It is the same with public spaces where local
authorities can enforce regulations. (see WHO pamphlet on Indoor air quality)
 Radon
Radon
In specific geographical areas, and in
buildings which have not been designed to be radon-proof,
concentrations of radon gas may reach levels that cause lung
cancer. The increased risk of cancer from radon exposure is
strongly enhanced for smokers, and several thousands of extra
cancers in Europe can be attributable to radon in regions of high
emission.
(see WHO pamphlet on Radon)
 Asbestos
Asbestos
As for radon, smoking substantially
increases the risk of cancer in asbestos-exposed populations
(see WHO pamphlet on Asbestos and health).
 Volatile organic compounds (VOCs)
Volatile organic compounds (VOCs)
VOCs originate mainly from solvents and
chemicals used at home or in offices. The main indoor sources
are: perfumes, hair-spray, furniture polish, glues, paints,
stains and varnishes, wood preservatives, pesticides, air
fresheners, dry cleaning, moth repellents and many other
products. The main health effects are eye, nose and throat
irritation. In more severe cases there may be headaches, loss of
coordination and nausea. In the long-term, some of them are
suspected to damage the liver, the kidneys and the central
nervous system as well as being potential carcinogens. Simple
measures, such as proper use of these products according to
manufacturers' instructions, and ventilating the rooms when using
such products, will keep the contamination at levels which can be
considered as safe for the health of most people. Acute
contamination will be prevented by the usual measures of home
safety regarding the use of chemicals.
 Tobacco
smoke
Tobacco
smoke
 Smoking generates a wide range of harmful chemicals
including nicotine, tar, formaldehyde, oxides of nitrogen and
carbon monoxide. It is now well known that smoking causes cancer.
It is also well known that passive smoking causes a wide range of
problems to the passive smoker ranging from being disturbed by
the smell, and eye, nose and throat irritation in children, to
cancer, increased risk of bronchitis and pneumonia, severe asthma
crises, and decreases in lung function. However, legislation
varies considerably between European countries. Other factors
such as the cost of tobacco have an obvious influence on the
number of smokers in countries. Local authorities should ban
smoking in all areas where they can: schools, parking lots,
cinemas, all municipal buildings and restaurants, with spaces
reserved for non-smokers when necessary.
Smoking generates a wide range of harmful chemicals
including nicotine, tar, formaldehyde, oxides of nitrogen and
carbon monoxide. It is now well known that smoking causes cancer.
It is also well known that passive smoking causes a wide range of
problems to the passive smoker ranging from being disturbed by
the smell, and eye, nose and throat irritation in children, to
cancer, increased risk of bronchitis and pneumonia, severe asthma
crises, and decreases in lung function. However, legislation
varies considerably between European countries. Other factors
such as the cost of tobacco have an obvious influence on the
number of smokers in countries. Local authorities should ban
smoking in all areas where they can: schools, parking lots,
cinemas, all municipal buildings and restaurants, with spaces
reserved for non-smokers when necessary.
| Consumption of Tobacco throughout the
European Community |
| Member
state |
%
adults smokers |
%male |
%female |
Per
capita consumption |
smoking
legislation |
| Austria |
30 |
40 |
21 |
1725 |
minimal
legislative control |
| Belgium |
25 |
31 |
19 |
1515 |
Complicated,
vague provision |
| Denmark |
42 |
45 |
39 |
1574 |
very
limited restrictions |
| Finland |
24 |
29 |
19 |
1613 |
widespread
ban |
| France |
34 |
40 |
27 |
2025 |
The
most comprehensive in the EU |
| Germany |
29 |
37 |
22 |
1939 |
No
national regulations |
| Greece |
37 |
46 |
28 |
3297 |
relatively
limited |
| Ireland |
30 |
28 |
32 |
2151 |
Fairly
widespread public ban |
| Italy |
32 |
38 |
26 |
1849 |
Mainly
public places and transport |
| Luxembourg |
27 |
32 |
26 |
|
relatively
widespread |
| Netherlands |
33 |
37 |
30 |
2465 |
complicated
provision |
| Portugal |
28 |
46 |
12 |
1969 |
similar
to Luxembourg |
| Spain |
36 |
48 |
25 |
2312 |
Extensive
ban in public places |
| Sweden |
23 |
23 |
23 |
1227 |
widespread
ban |
| United
kingdom |
28 |
29 |
28 |
1982 |
voluntary
code of practice |
 Formaldehyde
Formaldehyde
Formaldehyde comes mainly from particle
board, carpets and insulation foams. Beside eye, nose and throat
irritation, it may in some allergic people cause wheezing,
coughing, skin rash and other severe allergic reactions. High
concentrations may trigger attacks in people with asthma. The
measures that local authorities can take concentrate mainly on
avoiding the use of pressed wood products which contain urea -
formaldehyde resins.
 Biological pollutants
Biological pollutants
There is a wide range of biological
pollutants which includes pollen from plants, mites, pet hair,
fungi, parasites and some bacteria. Most of them are allergens
and can cause asthma, hay fever and other allergic diseases.
(See relevant pamphlets).
 Pesticides
Pesticides
As indoor air pollutants, pesticides are a
minor risk to health when used according to manufacturers'
instructions and when stored according to safety rules. (For
outdoor effects of pesticides, see
WHO pamphlet Pesticides and health). 
(2) Primary pollutants are those emitted
directly into the air, e.g. sulphur dioxide and volatile organic
compounds. Secondary pollutants are those formed by a
combination, or reaction (e.g. photochemical), with other primary
pollutants, e.g. ground level ozone.



Document Actions
Share with others