Androulakakis, A., et al., 2022, ‘Determination of 56 per-and polyfluoroalkyl substances in top predators and their prey from Northern Europe by LC-MS/MS’, Chemosphere 287, 131775 (https://doi.org/10.1016/j.chemosphere.2021.131775).
BMUV, 2022, Guidelines for PFAS assessment: recommendations for the uniform nationwide assessment of soil and water contamination and for the disposal of soil material containing PFAS, Bundesministerium für Umwelt, Naturschutz, nukleare Sicherheit und Verbraucherschutz (https://www.bmuv.de/fileadmin/Daten_BMU/Download_PDF/Bodenschutz/pfas_leitfaden_2022_en_bf.pdf) accessed 25 October 2022.
Brandsma, S. H. et al., 2019, ‘The PFOA substitute GenX detected in the environment near a fluoropolymer manufacturing plant in the Netherlands’, Chemosphere 220, pp. 493-500 (https://doi.org/10.1016/j.chemosphere.2018.12.135).
Bunting, S. Y., et al., 2021, ‘Emerging organic compounds in European groundwater’, Environmental Pollution 269, 115945 (https://doi.org/10.1016/j.envpol.2020.115945).
Cousins, I. T., et al., 2022, ‘Outside the safe operating space of a new planetary boundary for per-and polyfluoroalkyl substances (PFAS)’, Environmental Science and Technology 56(16), pp. 11172-11179 (https://doi.org/10.1021/acs.est.2c02765).
EC, 2022, ‘Proposal for a Directive of the European Parliament and of the Council amending Directive 2000/60/EC establishing a framework for Community action in the field of water policy, Directive 2006/118/EC on the protection of groundwater against pollution and deterioration and Directive 2008/105/EC on environmental quality standards in the field of water policy’, Brussels, 26.10.2022 COM(2022) 540 final 2022/0344 (https://environment.ec.europa.eu/publications/proposal-amending-water-directives_en) accessed 13 November 2022.
ECHA, 2022a, ‘Perfluoroalkyl chemicals (PFASs)’, (https://echa.europa.eu/hot-topics/perfluoroalkyl-chemicals-pfas) accessed 11 November 2022.
ECHA, 2022b, ‘Proposal to ban ‘forever chemicals’ in firefighting foams throughout the EU’, European Chemicals Agency’, (https://echa.europa.eu/de/-/proposal-to-ban-forever-chemicals-in-firefighting-foams-throughout-the-eu) accessed 5 September 2022.
EEA, 2019, Emerging chemical risks in Europe — PFAS, European Environment Agency (https://www.eea.europa.eu/publications/emerging-chemical-risks-in-europe/emerging-chemical-risks-in-europe) accessed 6 September 2022.
ETC/WMGE, 2021, Fluorinated polymers in a low carbon, circular and toxic-free economy, ETC/WMGE Report 2021/9, European Topic Centre on Waste and Materials in a Green Economy (https://www.eionet.europa.eu/etcs/etc-wmge/products/etc-wmge-reports/fluorinated-polymers-in-a-low-carbon-circular-and-toxic-free-economy) accessed 25 October 2022.
Gebbink, W. A. and van Leeuwen, S. P. J., 2020, ‘Environmental contamination and human exposure to PFASs near a fluorochemical production plant: review of historic and current PFOA and GenX contamination in the Netherlands’, Environment International 137(2020), 105583 (https://doi.org/10.1016/j.envint.2020.105583).
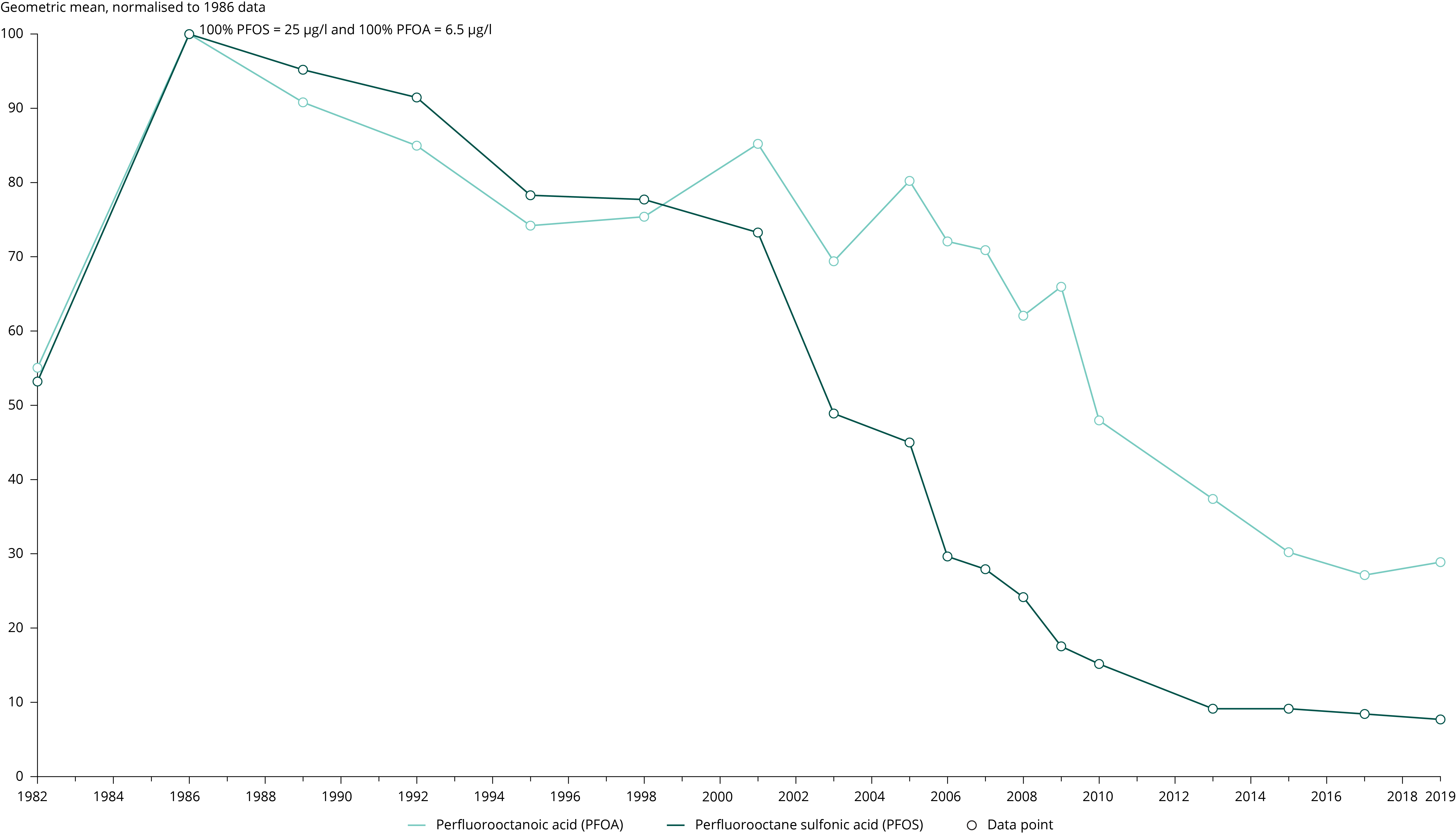
Göckener, B., et al., 2020, ‘Human biomonitoring of per- and polyfluoroalkyl substances in German blood plasma samples from 1982 to 2019’, Environment International 145, 106123 (https://doi.org/10.1016/j.envint.2020.106123).
Goldenman, G., et al., 2019, ‘The cost of inaction: A socioeconomic analysis of environmental and health impacts linked to exposure to PFAS’, Nordic Council of Ministers, (http://norden.diva-portal.org/smash/get/diva2:1295959/FULLTEXT01.pdf) accessed 11 November 2022).
Groffen, T., et al., 2017, ‘Perfluoroalkylated acids in the eggs of great tits (Parus major) near a fluorochemical plant in Flanders, Belgium’, Environmental Pollution 228, pp. 140-148 (https://doi.org/10.1016/j.envpol.2017.05.007).
HBM4EU, 2022, PFAS policy brief (https://www.hbm4eu.eu/wp-content/uploads/2022/06/HBM4EU_Policy-Brief-PFAS.pdf) accessed 29 September 2022.
Lobo Vicente et al., forthcoming, ‘HBM4EU results support the Chemicals’ Strategy for Sustainability and the Zero-Pollution Action Plan’,International Journal of Hygiene and Environmental Health, 247.
Lohmann, R., et al., 2020, ‘Are fluoropolymers really of low concern for human and environmental health and separate from other PFAS?’, Environmental Science and Technology 54(20), pp. 12820-12828 (https://doi.org/10.1021/acs.est.0c03244).
Miner, K. R., et al., 2021, ‘Deposition of PFAS “forever chemicals” on Mt. Everest’, Science of the Total Environment 759, p. 144421 (https://doi.org/10.1016/j.scitotenv.2020.144421).
Munoz, G., et al., 2015, ‘Spatial distribution and partitioning behavior of selected poly- and perfluoroalkyl substances in freshwater ecosystems: a French nationwide survey’, Science of the Total Environment 517, pp. 48-56 (https://doi.org/10.1016/j.scitotenv.2015.02.043).
Pramanik, B. P., 2015, ‘Occurrence of perfluoroalkyl and polyfluoroalkyl substances in the water environment and their removal in a water treatment process’, Journal of Water Reuse and Desalination 5(2), pp. 196-210 (https://doi.org/10.2166/wrd.2014.068).
RIVM, 2022, ‘PFAS restriction proposal’, Netherlands National Institute for Public Health and the Environment (https://www.rivm.nl/en/pfas/pfas-restriction-proposal) accessed 5 September 2022.
Reinikainen, J., et al., 2022, ‘The occurrence, distribution, and risks of PFAS at sites impacted by firefighting foams’, Science of the Total Environment 829, pp. 154237, (https://doi.org/10.1016/j.scitotenv.2022.154237).
Richterová, D., et al., 2023, ‘PFAS levels and determinants of variability in exposure in European teenagers–Results from the HBM4EU aligned studies (2014–2021)’, International Journal of Hygiene and Environmental Health, 247, pp.114057, (https://doi.org/10.1016/j.ijheh.2022.114057).
Sahlin, S., 2017, PFAS in the Baltic Sea region: inventory of awareness, actions and strategies related to highly fluorinated substances, PFAS, including PFOS,Policy Area Hazards, EUSBSR, Swedish Environmental Protection Agency (https://portal.helcom.fi/meetings/PRESSURE%207-2017-462/MeetingDocuments/7-7_Att%20Information%20on%20PFAS%20in%20the%20Baltc%20Sea%20Region.pdf) accessed 25 October 2022.
Serrano, L., et al., 2021, ‘Concentrations of perfluoroalkyl substances in donor breast milk in Southern Spain and their potential determinants’, International Journal of Hygiene and Environmental Health 236, p. 113796 (https://doi.org/10.1016/j.ijheh.2021.113796).
Schrenk, D., et al., 2020, ‘Risk to human health related to the presence of perfluoroalkyl substances in food, EFSA Panel on Contaminants in the Food Chain’, EFSA Journal, 18(9), pp. 6223 (https://doi.org/10.2903/j.efsa.2020.6223).
Schröter-Kermani, C., et al., 2013, ‘Retrospective monitoring of perfluorocarboxylates and perfluorosulfonates in human plasma archived by the German Environmental Specimen Bank’, International Journal of Hygiene and Environmental Health, 216(6), pp.633, (https://doi.org/10.1016/j.ijheh.2012.08.004).
Surma, M., et al., 2021, ‘Perfluoroalkyl substance contamination levels of pike (Esox luciusL.) and roach (Rutilus rutilusL.) from selected Masurian lakes in Eastern Europe’, Environmental Toxicology and Chemistry 40(12), pp. 3317-3327 (https://doi.org/10.1002/etc.5223).
Vito, 2022, Studie naar PFAS in lucht en deposities in de omgeving van 3M en Zwijndrecht, 2022/HEALTH/R/2680, VITO NV (https://www.vmm.be/publicaties/studie-naar-pfas-in-lucht-en-deposities-in-de-omgeving-van-3m-en-zwijndrecht#:~:text=3M%20en%20Zwijndrecht-,Studie%20naar%20PFAS%20in%20lucht%20en%20deposities%20in%20de%20omgeving,3M%2Dsite%20en%20de%20Oosterweelwerf) accessed 25 October 2022.
WHO Europe, 2017, Keeping our water clean: the case of water contamination in the Veneto region, Italy, World Health Organization Regional Office for Europe (https://www.euro.who.int/__data/assets/pdf_file/0019/341074/pfas-report-20170606-h1330-print-isbn.pdf) accessed 25 October 2022.
Weise, P., et al., 2022, Human Biomonitoring for Europe (HBM4EU)-first insights into the results of the initiative. Bundesgesundheitsblatt, Gesundheitsforschung, Gesundheitsschutz, 65(9), pp.936-939(https://doi.org/10.1007/s00103-022-03578-z).
Xu, Y., et al., 2021, ‘Serum perfluoroalkyl substances in residents following long-term drinking water contamination from firefighting foam in Ronneby, Sweden’, Environment international, 147, pp.106333 (https://doi.org/10.1016/j.envint.2020.106333)
Document Actions
Share with others